Ardelyx Provides Update on Growing Commercial Momentum and 2024 Strategic Priorities | Ardelyx, Inc.

Ardelyx on X: "Press Release: FDA Grants Appeal for Ardelyx's XPHOZAH® (tenapanor) Read more here: https://t.co/drMd3jYvKl https://t.co/DYmGNsa9Nt" / X

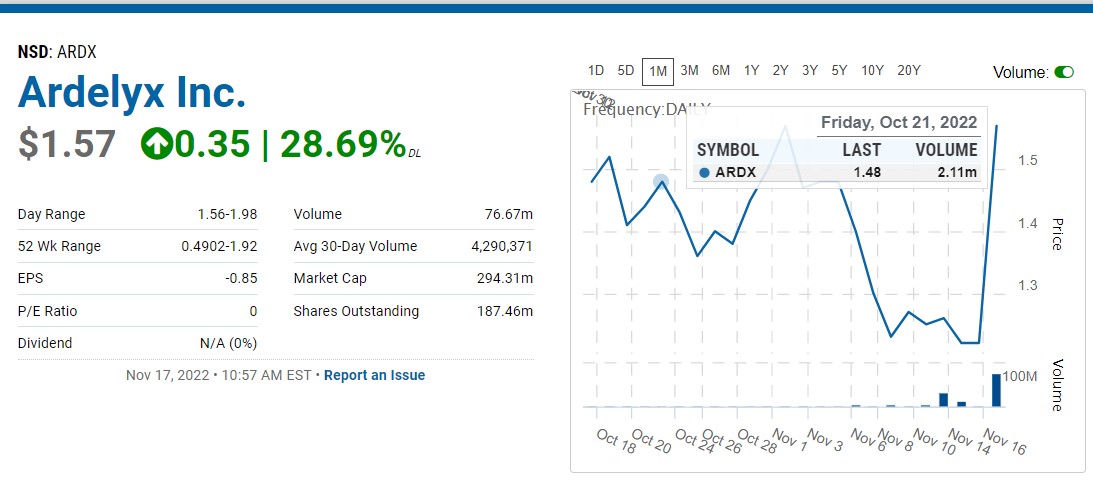
Ardelyx Inc Stock News | ARDX Company Stock News and Press Releases | FinancialContent Business Page

Ardelyx Announces Publication of 52-Week Phase 3 PHREEDOM Trial | PDF | Chronic Kidney Disease | Therapy